|
By Savannah Hari, QA/RA Specialist
« News & Press Releases
ISO/IEC 17025:2017 includes many changes. There are three main points to keep in mind: more options, involvement of risk, updates in current technology.
ISO/IEC 17025:2017 General requirements for the competence of testing and calibration was released in Nov 2017. All accredited labs have to move to the new standard by late 2020, three years after the final version was published. There are major differences with the formatting, phrasing, and numbering of sections from the 2012 standard. Reading through the 2017 edition and creating my own cross referencing table helped me find specific differences between the two versions.
The new standard offers more flexibility in the requirements for process, procedures, and organizational responsibilities. ISO 17025:2017 section 7.10 Non-conforming Work (previously 4.9) has been made more general.
Firstly, the title in the 2017 version is simply “Nonconforming Workâ€, wherein it was previously “Control of nonconforming testing and/or calibration workâ€. Management’s responsibilities towards nonconforming work are required to be defined, in comparison to the 2015 version where management must be designated and actions “including halting of work and withholding of test reports and calibration certificates†are defined.
There are also more options in the 2017 standard. Literally. Instead of having correlating sections to Corrective Action (4.11), Preventative Action (section 4.12), and Improvement (section 4.10), the new standard has Options A and B.
Option A addresses a management system that has control of records, improvements, corrective actions, internal audits, and management reviews, for example. Option B is more suited for a laboratory that fulfils the requirements of ISO 9001, “and at least the intent of the management system requirement specified in [Option A]â€. Option A includes a more detailed description of sections and their requirements.
Risk-based thinking is included throughout ISO 17025:2017. As stated in the standard, “this has enabled some reduction in prescriptive requirementsâ€. Option A includes a section specifying that the laboratory must: consider risks associated with lab activities; plan how to address them; and evaluate the effectiveness of these actions. Section 8.5 of Option A continues: “actions taken to address risks and opportunities shall be proportional to the potential impact on the validity of laboratory resultsâ€.
A formal risk management process is not required, however application of other guidances or standards is a possibility. Risk must also be considered with corrective actions in the 2017 standard. Risks determined during planning shall be updated, if necessary, when a nonconformity occurs.
Finally, risk has been incorporated into section 7.10 Nonconforming Work of ISO 17025:2017. Actions in response to a nonconformance “are based upon the risk levels established by the laboratoryâ€. Records of these actions must be retained.
ISO 17025:2017 addresses changes in technology made in the twelve years since the release of the second edition. Section 5.4.7 in 2005 has been expanded from “Control of data†to “Control of data and information managementâ€. It adds that the lab shall have access to the data and information needed to perform lab activities. 2017 also addresses off-the-shelf (OTF) software. Modifications to OTS software shall be authorized, documented, and validated before implementation. However, commercial OTS software that is not modified for use is not required to be validated.
Conclusion
A good place to start your update to ISO 17025:2017 is to look at options available, the incorporation of risk, and to review current software being used. Creating a cross reference table to track where sections have moved in the new standard will help later when you decide what changes need to be made in your QMS procedures. It takes careful reading and may be time consuming in the beginning, however it will ensure that small changes in the new edition are not missed.
anyone
Control of Calibrated Equipment Procedure [ISO 9001:2015]
The purpose of this procedure is to define your organization’s responsibilities and activities in order to ensure that all inspection and test equipment used for product, service and process verification is controlled and calibrated against nationally traceable standards at specified intervals and that such devices are available to ensure continuity of measurement capability.
Internal Audit Checklist [OHSAS 18001:2007]
The Occupational Health and Safety Assessment Series OHSAS 18001:2007 audit checklist will help ensure your audits address the necessary requirements. It stands as a reference point before, during and after the audit process and if developed for a specific audit and used correctly will provide the following benefits:
This OHSAS 18001:2007 audit checklist comprises tables of the certifiable (‘shall’) requirements, from OHSAS 18001:2007, each required is phrased as a question. This audit checklist may be used for element compliance audits and for process audits.
Gap Analysis Checklist [OHSAS 18001:2007]
The Occupational Health and Safety Assessment Series gap analysis checklist highlights the requirements contained in OHSAS 18001:2007.
After completing the Gap Analysis you will have a list of activities and processes that comply and ones that do not comply (GAPs).
Armed with this knowledge, it allows you to establish accurate budgets, timelines and expectations which are proportional to the state of your current management system when directly compared to the requirements of the standards.
Work Instuction Template
Work instructions are low-level documents showing operational detail. They are optional.
Work instructions might describe how to operate machinery, how to mix chemicals for a certain process, how to process a purchase order, or anything else that you feel is important enough to document and share.
Many companies avoid over-burdening their quality manual by allowing lower-level documents, such as procedures and work instructions to contain the operational detail. Then, simple reference is made to the procedures and work instructions from within the quality manual itself.
Product Realization & Planning Process
The purpose of this process is to define the activities required to ensure that product realization and planning encompasses the determination of product and process quality objectives, development of required processes, documentation and records.
Product quality objectives are defined in drawings and specifications, contracts, standards, samples, workmanship standards, and applicable legal and regulatory requirements.
Product realization planning includes the following:
• Definition and evaluation of production/service processes • Development of suitable and capable processes • Identification of special processes and consideration of associated risks and consequences • Development and implementation of appropriate process control measures • Development of instructions (where applicable) and training for process personnel • Identification of the records required to demonstrate product/process quality
Product realization plans are generated collectively by the Production/Service, Engineering and Quality Functions. The plans are defined in associated production/service documents (e.g. process flowcharts, production/service work orders, process control plans, operator/installation instructions, process validation reports, etc.).
Calibration Process
The purpose of this process is to ensure that all inspection and test equipment used for product and process verification is calibrated against nationally traceable standards at specified intervals and that such devices are available to ensure continuity of measurement capability.
Customer Satisfaction Process
This document describes the methods for measuring, monitoring and interpreting customer satisfaction to determine if you are meeting customer requirements.
Customer Complaints & Feedback Process
The purpose of this process is to moitor, measuring and take appropriate corrective action from customer complaints and feedback.
PDF samples [ISO 9001:2015]
PDF samples [ISO 14001:2015]
PDF samples from the Integrated Management System
Why reinvent the wheel?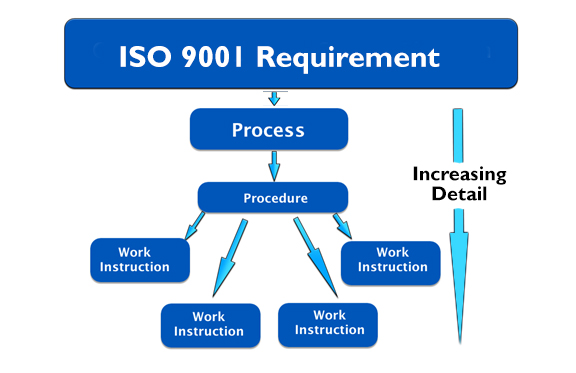
Our ISO 9001, ISO 14001 and OH&S 45001 templates are proven to work.
Ponga su cursor encima del globo para seleccionar la version en español.
ApplicationsManual
Explains the operational activities and responsibilities of ANAB and its customers (excluding forensics and management systems certification bodies).
Accreditation RequirementsIso 17025 Checklist Free
Defines accreditation requirements for ISO/IEC 17025 testing laboratories (non-forensic). To be used in conjunction with MA 2100 (see Manuals above). Additional supplemental program-specific documents may apply.
AR 2251, ISO/IEC 17025 Calibration Laboratories
Defines accreditation requirements for ISO/IEC 17025 calibration laboratories (non-forensic). To be used in conjunction with MA 2100 (see Manual above).
AR 2259, ISO/IEC 17025 Dimensional Measurement Laboratories (effective 2019/01/01)
Defines accreditation requirements for ISO/IEC 17025 dimensional measurement laboratories (non-forensic). To be used in conjunction with MA 2100 (see Manual above).
Supplemental Accreditation Requirements
SR 2401, Supplemental Accreditation Requirements: ANSI/NCSL Z540-1 Calibration Laboratories
SR 2402, Supplemental Accreditation Requirements: ANSI/NCSL Z540.3, Subclause 5.3, Calibration Laboratories
Defines supplemental requirements for accreditation and designation of testing laboratories under the Federal Communications Commission (FCC) Office of Engineering.
Defines supplemental requirements for accreditation of laboratories to the requirements of the EPA Energy Star program.
Defines supplemental requirements for accreditation of laboratories to the requirements of the DoD Advanced Geophysical Accreditation Classification Program (DAGCAP). Also see DoD information about this program.
Defines supplemental requirements for accreditation to Food Testing Laboratory Accreditation Program requirements for labs performing chemical and/or microbiological analyses in the examination of food products, ingredients in the production of food, in-process food samples, environmental samples pertinent to foods, and final products.
Defines supplemental requirements for accreditation to AAFCO Feed Testing Laboratory Accreditation Program requirements.
SR 2419, Supplemental Accreditation Requirements: TNI Non-Governmental Accreditation Body Environmental Laboratory Accreditation Program
Defines supplemental requirements for accreditation to the NELAC Institute (TNI) Non-Governmental Accreditation Body (NGAB) Environmental Laboratory Accreditation Program (ELAP) requirements.
Defines supplemental requirements for accreditation of laboratories to the requirements of the Department of Defense Environmental Laboratory Accreditation Program (DoD ELAP).
Defines supplemental requirements for accreditation of laboratories to the requirements of the EPA National Lead Laboratory Accreditation Program (NLLAP).
Defines supplemental requirements for accreditation of laboratories to the requirements of the EPA Toxic Substances Control Act (TSCA) Title VI Formaldehyde Program.
Defines supplemental requirements for accreditation to CPSC Testing Accreditation Program.
Defines supplemental requirements for accreditation of laboratories to the requirements of the VCCI Council Accreditation Program.
SR 2428, Supplemental Accreditation Requirements: Project 25 Conformity Assessment Program for Testing Laboratories
Defines supplemental requirements for accreditation of laboratories to the requirements of the Project 25 Conformity Assessment Program (P25 CAP).
Defines supplemental requirements for laboratories performing detection of suspect/counterfeit parts under the specific requirements of AS6171.
Defines supplemental requirements for horseracing laboratories accredited to ISO/IEC 17025.
Defines supplemental requirements for third-party telecom testing laboratories seeking NIST designation to Mexico. Shiva maha puranam in tamil pdf free download.
Defines supplemental requirements for accreditation of laboratories for the U.S Department of Energy Consolidated Audit Program (DOECAP).
Technical Accreditation Requirements
TR 2501, Technical Accreditation Requirements ISO/IEC 17025 Calibration Laboratories (Non-Forensic) - Scales and Balances - effective January 1, 2020
Administrative Process Rules
Applies to all customers and provides definitions and specific administrative process for accreditation statuses.
The assessment process determines the customer’s compliance with the requirements of the relevant accreditation standards, ANAB accreditation requirements, and any supplemental requirements, and the technical competence to the scope of accreditation. Various activities are used, both on-site and remotely, to evaluate conformance. Assessment activities include all locations where key activities of the customer are performed.
Defines the process when, for whatever reason, a customer delays the accreditation process during the application or assessment stage, and applies to all applicant and accredited customers.
Defines ANAB's process to determine, apply, manage, and accredit multiple-facility organizations (main, satellite, and temporary), mobile, and on-site conformity assessment services of the customer.
Defines ANAB's process when an accredited customer seeks to transfer accreditation to ANAB.
Defines assessment fees ANAB charges customers and the required travel requirements for ANAB assessors and technical experts.
Applies to all customers and stakeholders and provides administrative process rules for appeals and complaints.
Guidance Documents
Provides guidance on uncertainty requirements for ANAB-accredited laboratories that perform calibrations.
Establishes guidelines, clarifications, and recommendations for specific ISO/IEC 17025 requirements, and specific policies and instructions as defined by ANAB.
GD 2701.01, Guidance on Corrective Action and Cause Analysis (presentation)
Provides guidance on interpolation and extrapolation of calibration data as related to calculating measurement uncertainties and the expression of those uncertainties on the customer’s scope of accreditation and any accredited calibration or testing reports.
GD 2702.01, Guidance on Interpolation and Extrapolation of Calibration Data (presentation)
Provides general accreditation guidance for accredited customers to purchase calibrations that are fit for their intended purpose.
Scopes of Accreditation
To assist in drafting scopes of accreditation and to clarify ILAC guidance documents and ANAB requirements, while helping standardize formats across the range of potential ISO/IEC 17025 accredited tests.
FM 2850, ISO/IEC 17025 Testing Scope Template (contact ANAB)
FM 2864, ISO/IEC 17025 FCC Testing Scope Template (contact ANAB)
PR 2351, Preparing a Draft Scope of Accreditation for ISO/IEC 17025 Calibration Laboratories
To assist in drafting scopes of accreditation and to clarify ILAC guidance documents and ANAB requirements, while helping standardize formats across the range of potential accredited calibrations in the sphere of ISO/IEC 17025 accredited operations.
FM 2880, ISO/IEC 17025 Calibration Scope Template (contact ANAB)
To assist in drafting scopes of accreditation and to clarify ILAC guidance documents and ANAB requirements, while helping standardize formats across the range of potential ISO/IEC 17025 accredited dimensional measurements.
FM 2882, ISO/IEC 17025 Dimensional Measurement Scope Template (contact ANAB)
Other Documents
FM 2804, Technical Competence Evaluation (OPIEF) (contact ANAB)
Iso 17025 Manual Free
FM 2807, Traceability and In-house Calibration Tracking (contact ANAB)
CL 2900.01, ISO/IEC 17025:2005 General Accreditation Requirements Checklist (contact ANAB)
CL 2900.04, ISO/IEC 17025:2017 General Accreditation Requirements Checklist(contact ANAB)
Change Notifications
Used by customers to notify ANAB of any organizational changes, such as change of name, ownership, location, contact information, management, key personnel, accounting information, unsatisfactory PT/ILC, or scope of accreditation.
ISO/IEC 17025:2017 – General requirements for the competence of testing and calibration laboratories has been released. This standard specifies the general guidelines for the competence, impartiality, and consistent operation of laboratories.
In laboratories, instrument testing and calibration is integral for maintaining accuracy, as any factors that can attribute to inaccurate measurements need to be limited so the instruments can reliably carry out their very purpose. Assuredly, ISO/IEC 17025 has become the international reference for testing and calibration of laboratories desiring to demonstrate their capacity to deliver trusted results. In a way, it is the reliable source for providing reliability.
The previous version of ISO/IEC 17025 was the second edition, published back in 2005. Since then, market conditions have changed. Furthermore, the shared aspects among ISO international standards has driven the need for further harmonization among other existing international documents.
In meeting these two progressions, the third edition of the standard, ISO/IEC 17025:2017, has undergone numerous changes. Developed jointly by ISO and IEC in the Committee on conformity assessment (CASCO), the process approach of the document now matches that of newer standards such as ISO 9001, ISO 15189, and ISO/IEC 17021-1.
In fact, ISO/IEC 17025:2017 specifically describes the commonalities with it and ISO 9001:2015. The updated standard for testing and calibration laboratories has also adopted a new structure to align it with the other existing ISO/IEC conformity assessment standards, such as the ISO/IEC 17000 series for conformity assessment.
The above changes to the international standard are focused on trends with ISO documents. As for meeting the current industry needs, the changes to ISO/IEC 17025:2017 include:
ISO/IEC 17025:2017 is also available as part of the following standards packages:

ISO/IEC 17025:2017 – General requirements for the competence of testing and calibration laboratories is available on the ANSI Webstore.
SAMPLE PROCEDURE PROCEDURE 1.0 Responsibility 1.1 Purchasing and Quality share responsibility for the qualification and monitoring of suppliers. 2.0 Qualification and Rating of Suppliers 2.1 Suppliers' quality capabilities are evaluated through the use of Supplier Self-Surveys and where necessary, on-site audits, before orders for supply of materials and services are placed. The effectiveness of the suppliers' quality system shall be reviewed at intervals consistent with the complexity of the items supplied and the suppliers' performance. 2.2 Quality and delivery performance of all suppliers is continuously monitored via a supplier performance rating system and a monthly supplier performance report is distributed to all concerned functions. Suppliers with inadequate performance are asked to implement corrective actions and submit a written response. Suppliers that demonstrate inadequate performance and show no sign of improvement are removed from the Approved Supplier List and discontinued from use. Iso 17025 Procedures Templates Free3.0 Approved Supplier List 3.1 The Quality Manager maintains an Approved Supplier List which includes all suppliers whose supplies/processes affect the quality of the products/services, and who have been surveyed/audited and have demonstrated by performance, their ability to meet the specified quality requirements. Orders may only be placed with suppliers that are on the list. Emergency purchases from suppliers that are not on the list are allowed with written permission from the Purchasing Manager and the Quality Manager. An updated Approved Supplier List is printed and distributed monthly. 3.2 A file, which includes all qualification documentation and performance records, is maintained on each supplier. 4.0 Purchasing Data 4.1 All purchasing documents are prepared by Purchasing. The documents clearly describe ordered products/services. They include precise identification of the products/services, reference applicable standards and other relevant technical data, and state quality and compliance requirements. All purchasing documents are reviewed and approved by the Purchasing Manager prior to release. 5.0 Verification of Purchased Product 5.1 In the event Quality determines that a verification of purchased products will be required at the supplier's facility, verification arrangements will be specified on the Purchase Order. 5.2 Our customers are given the right to verify purchased products at our supplier's premises. In this case, XYZ Company is not relieved of the responsibility for products meeting stated quality requirements. 6.0 Settlement of Quality Disputes 6.1 The Quality Manager and Purchasing have the responsibility and authority to settle all disputes with suppliers regarding the quality of their material/products/services or matters such as inspection and testing methods. Open communication channels with all approved suppliers will be maintained to provide for the quick resolution of quality disputes. In the event the Quality Manager and Purchasing cannot resolve a dispute, he or she will request the assistance of the General Manager. Disputes involving the rejection of material/products from a supplier shall be documented. Comments are closed.
|
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
